Investigation: Why Are Cells So Small?

Essential Question: How does the size and shape of a cell influence the speed at which materials can move into and out of the the cell?
Process: Create cell models using agar molds to compare rates of diffusion.
Materials:
Agar Mold with Bromothymol Blue (made in advance)
Tweezers, Scalpel (or plastic knife)
Ruler, Beaker with white vinegar
Procedure
1. You will receive a small tray filled with an agar mold. *See below for directions* Avoid handling the agar with your bare hands and use a scalpel and tweezers to cut three agar cubes with the following approximate dimensions. Save your agar, you will need it later!
1 cm x 1 cm x 1 cm (small)
2 cm x 2 cm x 2 cm (medium)
1 cm x 1 cm x 6 cm (large)
2. Measure your cubes (the actual dimensions may not be perfect, depending on how you cut it) and determine the surface area, the volume, and the SA:V ratio. Record on data table.
3. Drop each block into a separate beaker (or container) of vinegar. The agar has been infused with a chemical called bromothymol blue, the blue will turn to a yellow in the presence of acid. You will be able to observe this change with your cubes. Record the time it takes for the blue to completely disappear.
| Actual Dimensions | Surface Area | Volume | SA / V | Time (Blue to Orange) | |
| Small Cube | |||||
| Medium Cube | |||||
| Large Cube |
Part 2: How Does Shape Influence Rates of Diffusion?
With what remains of your agar, design a cell that maximizes volume and mass, but minimizes diffusion time.
Your "cell" will compete with other cells in the class to see which one has the fastest diffusion time.
Rules:
- No donut-like holes through the agar cell - cell membranes cannot sustain this shape
- No poking or agitating the beaker when the cell is submerged
- Instructor determines when 100% diffusion has occurred
- Agar cell will be massed at the end of the race
- Winner = highest ratio of mass divided by time
Sketch your design below.
Analysis
1. Which of the initial cubes had the fastest diffusion time? Which had the slowest?
2. Which of the three variables you tested seemed to have the biggest impact on the rate of diffusion? Explain how you know this.
3. How does the agar cube model the cell and the cell membrane?
4. What designs (Part 2) seemed to have the fastest diffusion rate?
5. How do these experiments model the cell and the cell membrane?
Agar Recipe:
15 g of agar in 1 liter water (or follow directions on packaging). You do want the agar to be thick so that it can be handled, so reduce water amounts. Agar is boiled in DI water and then allowed to cool. Knox gelatin can also be subsituted, but you may need to play around with the measurements.
While it is cooling, add .1 g of bromothymol blue (or about 10 ml aqueous solution, you just need to ensure that the agar turns a dark blue.) If the mixture is green/yellow, then add NaOH until it turns blue.
Pour agar into trays for students. You can be creative with the trays (ziploc tupperware containers should work, or even metal dissecting trays.) The molds must be at least 2 cm deep. Molds can be covered and refrigerated.
These are the molds I created using specimen containers and the lids from a box of micropipettes. You can be creative!

Alternatively, you can add phenolphthalein to the agar and then submerge cubes in sodium hydroxide. <-- this tends to be more expensive than white vinegar, and NaOH is dangerous to handle.
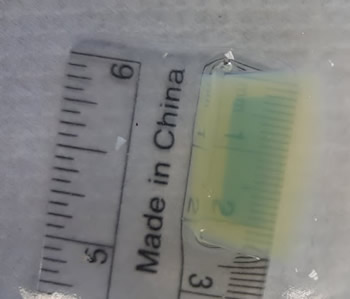
Image below shows saturation of agar by vinegar. The yellow area started as blue. A ruler placed under the flask can be used as another way to measure the rate of diffusion.
Other Activities on Cell Transport
Observe Diffusion in a Bag – model diffusion using a plastic baggie, iodine and a beaker. This article explains what happens.
Transport Across the Cell Membrane – simple diagram shows how molecules enter the cell through diffusion, facilitated diffusion, and osmosis
Cell Membrane Images – work in groups to create captions and titles for images depicting the cell membrane and transport across it.
Case Study: Cystic Fibrosis – for AP Biology, examines the role of cell membrane proteins in clearing mucus from the lungs.
Observing Osmosis – use an egg, vinegar, corn syrup, will take a few days
Salt and Elodea – quick lab to observe the effects of salt water on elodea cells

