
Nasonia Genetics - A Jewel of a Wasp
Resources:
Objective: In this lab you will explore the genetics of Nasonia vitripennis, or the jewel wasp, to determine how a trait is inherited. You will cross a P (parent) generation and determine from the numbers and types of offspring, how the gene coding for eye color is inherited.
Nasonia exhibit negative geotaxis and can also be used in animal behavior labs.
Introduction to the Jewel Wasp
Nasonia are excellent organisms for research and teaching. These parasitoid wasps have been the subject of genetic, ecological, evolutionary and developmental research for over 40 years. Two general features that make these insects such excellent study organisms are (a) ease of handling and rearing, and (b) interesting and diverse biology. Nasonia are readily reared on commercially available fly pupae (the hosts).
Virgin females and males are easily collected in the pupal stage (there is a 3 day time window for virgin collection). Adults are user friendly and can be handled without the need for anaesthetization. Nasonia has a short generation time (two weeks), but can be stored under refrigeration for periods of time, allowing for flexibility in experimental timing.
A key feature of Nasonia is haplodiploid sex determination; males are haploid and develop from unfertilized eggs and females are diploid and develop from fertilized eggs. Nasonia are small parasitoid wasps (Hymenoptera: Pteromalidae) that sting and lay eggs in the pupae of various fly species, primarily blowflies and fleshflies.
Life Cycle
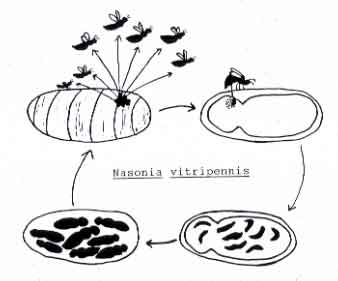
When a female encounters a host puparium, she first examines the host, then drills through the host puparial wall with her ovipositor. She injects venom into the pupa, which will eventually kill the fly. The female then commences laying eggs upon the host, underneath the puparial wall. She typically lays from 20 to 50 eggs per Sarcophaga bullata pupa. The female may lay these eggs in one bout or may take a number of hours to complete oviposition.
The female also uses excretions from her ovipositor to construct a feeding tube from the pupa to the puparial wall. From this she feeds on host hemolymph, which appears to be important in the production of additional eggs. At 25 C, eggs hatch around 36 hours after being laid. Developing larvae complete 3 instars and then pupate within the host around 9 days after laying.
Pupal development takes approximately 3 days at which time the pupa go through three visible stages: whites, black and whites, and blacks. When the pupa looks almost black, you know that your adults are soon to emerge. Male and female pupae can easily be distinguished during this time. Adults eclose from pupation within the host, and then chew an exit hole. Emergence typically occurs by 14 days.
At the black and white stage, you can also distinguish male from female wasps, at this time it is best to separate them to insure virgin females for later crosses. Mating occurs immediately upon emergence from the host. Courtship behavior is brief (typically taking 1-2 minutes) and involves stereotypic courtship displays. After mating, females disperse from the natal patch in search of new hosts.
Genetics
In many respects, Nasonia is a superior organism for genetic research. The important features that make it so are (a) short generation time (b) large family sizes, (c) ease of handling (including virgin collection), (d) ability to inbreed and produce healthy lines. Nasonia is particularly suited for the study of complex genetic traits, due to advantages provided by haploid males and the ability to easily produce inbred lines and genetically identical recombinant individuals. All three species of Nasonia have 5 chromosomes, corresponding to 5 linkage groups. currently there are about 20 mutant strains available, most of which are eye color, body color, morphological and embryonic lethal mutations. The mutant strain you'll be studying causes the normally black eyes (wild type) to be red (mutant)
Handling and Rearing
Stock Maintenance: Stocks are easily maintained in Nasonia in plastic or glass vials or test tubes. Emerging females are collected into a new vial by placing the vial over the original vial with emerged wasps. Females are negatively geotaxic and move into the new vial. Hosts are then placed into the new vial (usually approximately 1 - 2 wasps per host). Fourteen days later (at 25 C) the next generation emerges. It's as simple as that. No special feeding or handling is necessary.
Stocks can be slowed down by placing them at cooler temperatures, or speeded up (up to about 28 C). Cultures can be placed under refrigeration for a couple of weeks if necessary. This is best done at the yellow pupal stage and adult stage, but can be done at other life stages as well. Adult females can also be kept alive for several weeks at 250 C with a small amount of honey, and females can live for over a month if provided with fresh hosts
Collecting Virgins: Virgin collection is very easy in Nasonia. Wasp pupae can be sexed in the pupal stage, which provides a three day time window for virgin collection. They are immobile in the pupal stage, and therefore can be collected without the need for anesthetization. Individuals are most easily sexed in the black pupal stage, but with minimal training can be readily distinguished as yellow pupae. One looks for the presence of an ovipositor in the distal end of the abdomen. In N. vitripennis, males can also be distinguished by small wing pads.
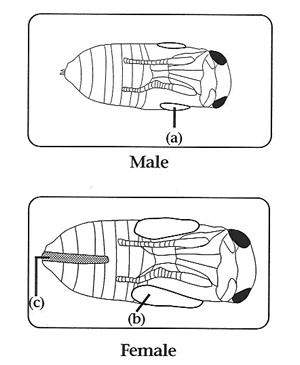
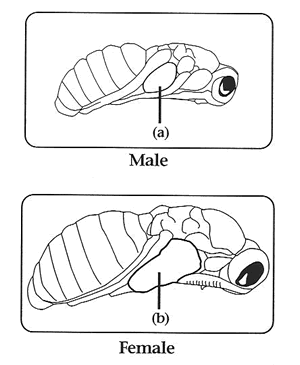
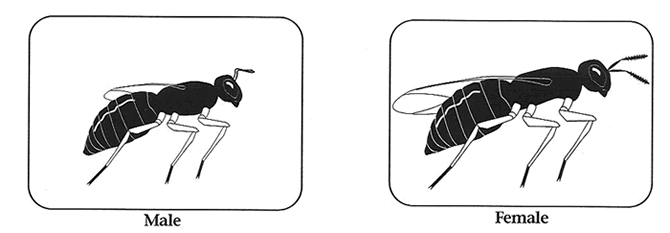
Handling Adults: Adults are very user friendly and can be sorted and used in experiments without anesthetization. Although females can fly, they do not do so readily. However, they are positively geotactic. Therefore, to set up females individually on hosts, one need only dump a few females onto a surface and then place test-tubes over the crawling individuals.
They will then conveniently climb into the tube. Add a host (or two) and plug the tube with cotton and you are done. Large numbers of individuals can be efficiently handled in this way. Females can easily be distinguished from males in the adult stage, as they are larger and their wings extend beyond thier abdomen. Females also have a darker colored body and legs, and a thicker antennae. Males have a greenish color and light colored thin antennae.
Collecting Eggs: The easiest way to collect eggs is to allow females to lay eggs for a prescribed period of time on a host to which their access is restricted to one end. This is accomplished by placing the host into a foam plug with a hole in one end, and placing this with the female into a test tube. After an oviposition period (the narrower the time, the more synchronized the eggs), hosts are removed, the puparial end is popped off with a probe, and eggs are collected with a fine brush. For maximum egg production, it is recommended that females be allowed to host feed for 2 - 3 days prior to placing them onto plug hosts for egg collection.
Euthanasia: When you are finished studying the wasps, subjects can be easily disposed of by placing them in the freezer for two or more hours. It is best to not release wasps into the environment, as mutant strains may have a negative impact on local species.
Procedure
1. Obtain a test tube of dark-eyed males and a test tube or red-eyed females.
2. Tap each tube gently onto the table. The Nasonia will fall to the bottom. If some of them get caught in the cotton, simply twist the cotton a little to dislodge them.
3. Once the Nasonia are in the bottom of the tubes, removes the contton plugs and invert the tube with the females over the tube with the males. Make sure the lips of the test tube line up so the wasps don't escape.
4. Gently tap the tubs on the table of flick the top tube so the female Nasonia fall down into the tube with the males.
5. Recap the tube containing the Nasonia with the cotton plug. Label the tube with your name(s) and place in rack.
6. Incubate your tube at room temperature for 24 hours. This will allow the males and females to mate.
7. Obtain two sarcophaga hosts from your teacher.
8. Remove your test tube from the rack. Tap the tube on the table so the male and female nasona fall tothe bottom.
9. Remove the cotton plug and place two hosts into the test tubes.
10. Replace the cotton plug and return the tubes back to the rack to incubate.
11. To insure that the female Nasonia parasitize the hosts, leave the Nasonia in the test tube for at least four or five days. After that time, remove the parental generation and place them in the morgue.
12. Depending on conditions, the F1 Nasonia should begin to emerge from the hosts in 14-15 days.
13. Once all the F1 Nasonia have emerged, they need to be euthanized. Place them in the freezer for two hours to euthanize them.
14. After euthanization, gently pour your F1 Nasonia onto a tray on your table. Use a paintbrush or other utensil to separate them into red-eyed females, dark-eyed females, red-eyed males, and dark-eyed males.
15. Keep a tally of your results, you may want just a rough draft. Later you'll need to organize this onto a chart for your lab report.