How Wood Alcohol Poisonings Advanced Toxicology
 In the days before Prohibition, New York was trying to update it's coroner's office by improving laboratory practices and using science to determine the cause of death and solve crimes. Up until 1917, the coroner was an elected official, and one who had no science or medical background. Murders of all kinds were written off as suicides, or "acts of God." Poisons had become a popular way to rid oneself of a rival or family member because it was unlikely that its effects would be detected.
In the days before Prohibition, New York was trying to update it's coroner's office by improving laboratory practices and using science to determine the cause of death and solve crimes. Up until 1917, the coroner was an elected official, and one who had no science or medical background. Murders of all kinds were written off as suicides, or "acts of God." Poisons had become a popular way to rid oneself of a rival or family member because it was unlikely that its effects would be detected.
In 1918, Charles Norris was appointed as the Chief Medical Examiner and was a pioneer in forensic toxicology. He, in turn, hired a chemist named Alexander Gettler who was given the task of developing methods to determine if poisons were the cause of death. Gettler spent time grinding up livers and testing a variety of substances. In the years leading up to the Prohibition, Gettler noted a rise in the number of deaths attributed to wood alcohol poisoning.
------- Excerpt from the Poisoner's Handbook -------
Wood alcohol -- technically known as methyl alcohol, but also as wood spirit, hydroxymethane, carbino, colonial spirit, Columbian spirit, and, some years later, methanol -- was in itself nothing new. The ancient Egyptians had used it in their embalming processes. For centuries it had been the essential ingrediant in homemade whiskey. Its chemical formula had been identified in 1661 by a chemist who called it "spirit of box" because he'd made it by distilling boxwood. The term, methyl was derived from the Greek methy (meaning wine) and hyle (meaning wood, or more precisely, path of trees).
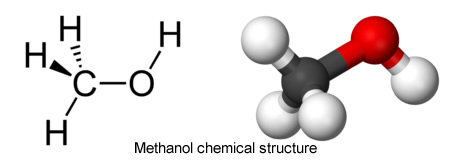
The chemical structure of wood alcohol is simple: three hydrogen atoms bonded to a single carbon atom (in a cluster known as a methyl group), with one oxygen atom and another hydrogen atom tagging along. It is also simple to make, as industrialists and moonshiners had realized, requiring little more than wood and heat. The process was called destructive distillation. Slabs and slices of wood went into a closed container and were heated to at least 400 degrees Fahrenheit (204 degrees Celsius). As the wood cooked into charcoal, its natural liquids vaporized. The vapor could be cooled, condensed, and distilled into a rather murky soup containing methyl alcohol, acetone, and acetic acid. A second distilling would separate out the pure methyl alcohol, a liquid as clear as glass and as odorless as ice, from the other ingredients.
-------------------------------------------------------------------------
"Industrial" alcohol was basically grain alcohol (ethanol) with other chemicals mixed in to make it undrinkable, methanol was one of the common additives. The adding of these chemicals was required by law after Prohibition to prevent industrial alcohol from being used as a beverage. Basically, the government ordered the alcohol to be laced with chemicals that would make it undrinkable. As a result, there were many bootleggers who attempted to make industrial alcohol drinkable and less toxic by redistilling, diluting or mixing it with other chemicals. None of these procedures was particularly effective, and people who chose to drink alcohol illegally would be risking their lives.
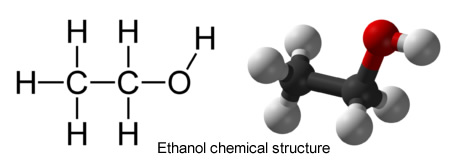
Why is methyl alcohol so dangerous when other acoholic beverages are not? Liquors usually contain about 3% ethanol which is often produced from fermenting grains, fruits, and vegetables. In a way to replace now banned liquors during the era of Prohibition, people turned to questionable distillation methods and drinks that were a mixture of ethanol and methanol. Even small quantities of methanol can be fatal, but the risk did not deter some people.
Methanol is toxic when ingested, inhaled or even absorbed in the skin. Methanol , when ingested, breaks down into even more toxic substances, a process called toxification. Enzymes in the liver first convert methanol to formaldehyde which is then converted to formic acid. This process takes up to 30 hours from the initial exposure to methanol, and means that you might not die initially from its consumption, but may experience symptoms a day later. Usually the first sign of methanol poisoning is loss of vision. If the person recovers from the toxins, the blindness is permanent as formic acid actually damages the optic nerve.
As Gettler had predicted, the number of cases of poisoning increased. In 1926, in New York City alone, 1200 became sick by poisonous alcohol and 400 died. Bootlegging of alcohol, much of which contained methanol continued, poisonings became a public health issue. Gettler continued to work these cases, publicizing deaths by alcohol to raise awareness of the problem. In 1933, the 18th amendment was repealed and the program that required industrial alcohol to be made toxic was also ceased.
____ 1. According to the article, which of the following is true? (Standard 1: Cite text evidence)
a) children were most likely to suffer from methanol poisoning b) the Prohibition legalized the use of methyl alcohol
c) the structure of methanol and ethanol is identical d) methyl alcohol is toxic and can cause blindness
____ 2. Select the sentence the supports the central idea of the article. (Standard 2: Central ideas)
a) Poisons had become a popular way to rid oneself of a rival or family member because it was unlikely that its effects would be detected.
b) In the years leading up to the Prohibition, Gettler noted a rise in the number of deaths attributed to wood alcohol poisoning.
c) In 1918, Charles Norris was appointed as the Chief Medical Examiner and was a pioneer in forensic toxicology.
d) Liquors usually contain about 3% ethanol which is often produced from fermenting grains, fruits, and vegetables.
____ 3. What best describes the structure of the article? (Standard 2: Central ideas)
a) a description of how medical examination changed in the 1900s
b) a persuasive essay on why the Prohibition was not an economically sound idea
c) a comparison of methyl alchohol to ethyl alcohol and a narrative about why methanol is toxic
d) an opinion piece about what life was like during Prohibition
____ 4. Why did it take so long for a person to die after ingesting a lethal dose of methanol? (Standard 1: Cite text evidence)
a) it took a while for the poison to circulate throughout the body and reach the brain
b) the poison accumulated in the tissues of the person and sometimes took years to kill them
c) only some people are sensitive to methanol
d) methanol is converted by the liver to an even more toxic substance (toxification)
____ 5. What is the relationship between prohibition and methanol? (Standard 5: Relationships)
a) prohibition caused methanol to be made illegal
b) prohibition resulted in more methanol poisonings
c) prohibition reduced the number of alcohol related deaths
d) prohibition advanced the field of toxicology
____ 6. Which of the following is the best definition of "toxification"? (Standard 4: Meanings)
a) a process where one substance is converted into another substance
b) a series of chemical reactions that occur within the blood
c) a process where one substance is converted into another substance that is more toxic than the first
d) a procedure where a scientist determines how potent a toxin is
____ 7. Which of the following data supports Gettler's prediction that methanol poisonings will increase after prohibition? (Standard 8: Reasoning)
a) a comparison between the number of deaths from methanol poisoning before and after prohibition went into effect
b) a toxicology report on how quickly rats succumb to methanol poisoning
c) a listing of how much alcohol containing methanol was confiscated
d) hospital records showing how many people had been treated for alcohol related illnesses
____ 8. Complete this analogy: Ethanol is to grain as methanol is to ... (Standard 4: Meanings)
a) poison b) wood c) fruit d) blindness
____ 9. There are two potential sources for a person to acquire methanol, what are they - circle 2? (Standard 4: Meanings)
a) it can be directly distilled from burning wood
b) it can be harvested from the organs of someone who has died from methanol poisoning
c) it can be fermented using rotting fruit
d) it can be acquired from industrial alcohol that has been contaminated
10. In your own words, describe the difference between a molecule of ethanol and a molecule of methanol. (Standard 8: Visual Information)


